Gain’s Clinical Candidate GT-02287
Earlier this month, Gain Therapeutics announced the dosing of the first two subjects in a Phase 1 clinical trial of GT-02287, a molecule discovered and developed by Gain for the treatment of neurodegenerative diseases. GT-02287 is a small molecule that binds and stabilizes an enzyme called GCase, increasing its activity in the cell. GCase is responsible for clearing toxic substrates in the lysosome, but when GCase levels are lowered, such as in some neurodegenerative diseases, these substrates accumulate and lead to the build-up of neurodegenerative disease, such as GBA-Parkinson’s disease. GBA-Parkinson’s is a genetically defined subpopulation of Parkinson’s characterized by the lack of functional GCase. By increasing the activity of GCase, GT-02287 has shown preclinically, the ability to deplete toxic substrates in the cell and have a positive impact on disease. One of these toxic substrates is alpha-synuclein, a neuronal protein that is considered to be the hallmark of Parkinson’s disease. Depletion of alpha-synuclein has been a goal of therapeutics in the Parkinson’s space for years, and Gain’s GT-02287 has shown great promise in its ability to deplete this substrate and affect Parkinson’s disease progression. While it remains an important target for the treatment of Parkinson’s disease, alpha-synuclein is the cause of many other neurodegenerative diseases known as synucleinopathies.
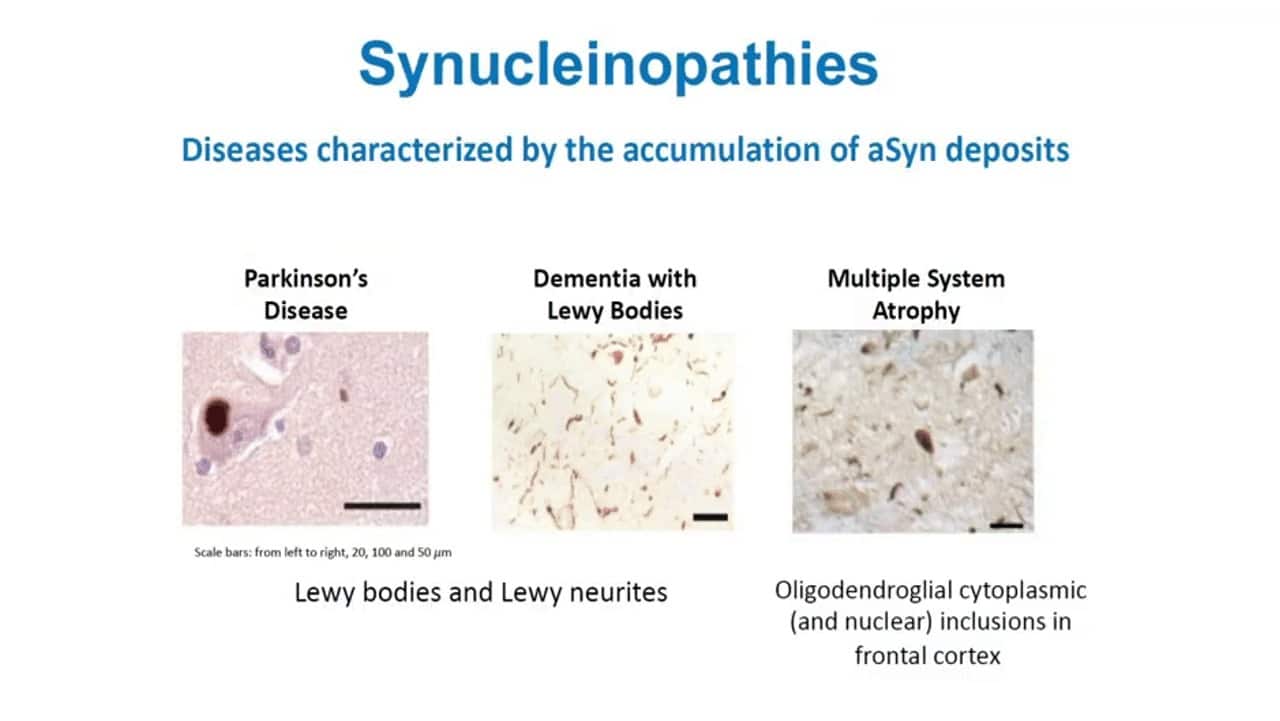
The Synucleinopathies
Synucleinopathies such as Parkinson’s disease (PD), Dementia with Lewy Bodies (DLB), and Multiple System Atrophy (MSA) are neurodegenerative disorders that present with a myriad of movement and cognitive abnormalities. Although somewhat similar, all three have clinical symptomatology, pathology, and outcomes that differentiate into distinct disorders. PD presents primarily as a movement disorder, whereas MSA presents as a balance and coordination disorder with a more rapid clinical course. DLB primarily begins as a memory and cognition disorder with associated movement abnormalities later in the disease course. Despite their distinct phenotypes, all three disorders result from the accumulation of misfolded alpha synuclein, a protein that facilitates signal transmission between neurons by regulating neurotransmitter release. Misfolded alpha synuclein interacts with and misfolds the normal, endogenous alpha synuclein, continuing a chain reaction of misfolding. Accumulation of misfolded alpha synuclein ultimately leads to neurodegeneration and disease symptoms. Due to the importance of alpha-synuclein in neurological disorders, several therapies are in development with the goal of targeting this protein.
Therapeutic Interventions
A popular therapeutic mechanism is utilizing immunotherapy. Many companies are developing engineered antibodies, specific to alpha-synuclein, that would in theory neutralize the toxic forms of the protein, and potentially delay the progression of disease. While these therapies have yet to be validated clinically, a key limitation of this approach is the focus on a specific substrate. These therapies can deplete the toxic forms of the protein that aggregate, but are unlikely to have an effect on any of the precursor proteins in the disease cascade. Gain’s approach and clinical candidate GT-02287 is unique and differentiated in that it intervenes at the start of the disease cascade. By targeting an enzyme at the start of the disease cascade, GT-02287 has shown to have a positive effect on all subsequent disease-causing events, including alpha-synuclein. GT-02287 is the first small molecule approach to show effects at all levels of the disease cascade and due to its strong effect on alpha-synuclein, has potential applications in Parkinson’s disease and other synucleinopathies.